 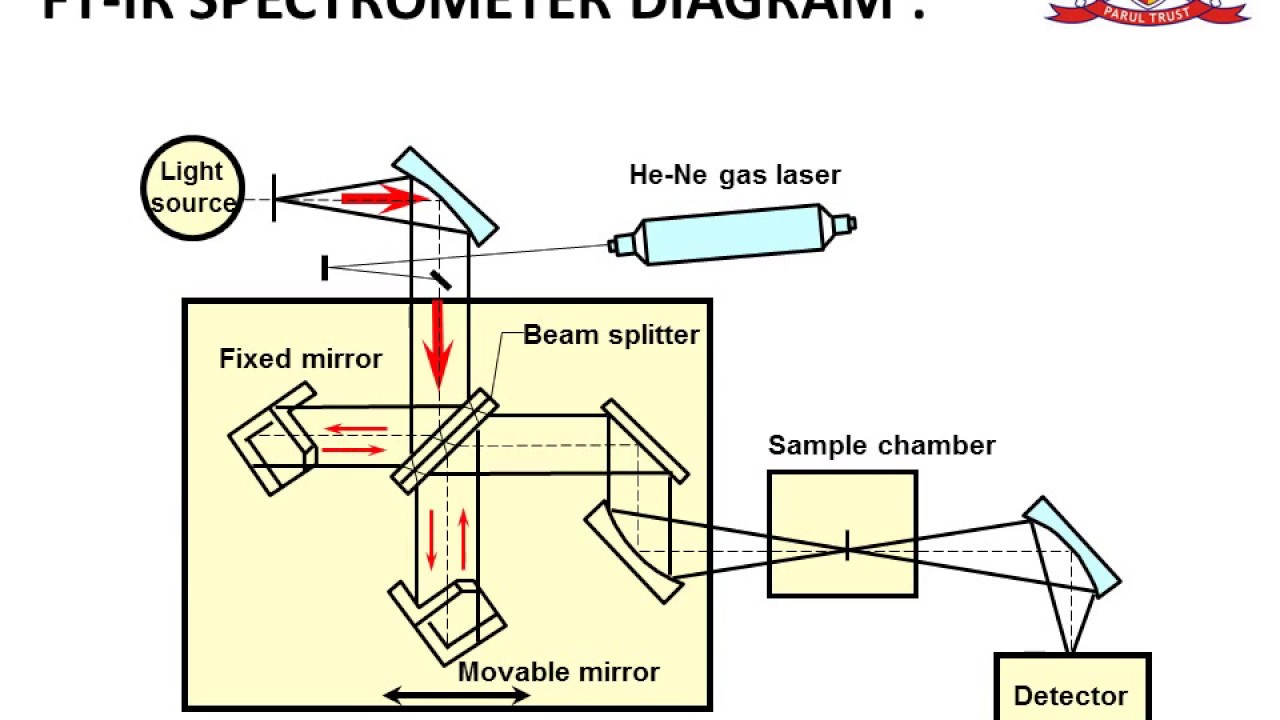
For this reason, Raman spectroscopy and IR spectroscopy tend to be complementary techniques, as they provide different spectra depending on the chemical bonds present in the sample. light is absorbed due to change in dipole during vibration), or both. light is scattered due to change in polarizability during vibration), IR active (i.e. While the full scope of such vibrational modes is vast, the vibrational modes can either be Raman active (i.e. In inelastic collisions between infrared light and molecules (either at the surface of a solid material or as a gas or liquid) some collisions produce characteristic vibrations of varying modes dependent on the nature of the bond itself, but with a characteristic vibrational frequency. The basic premise of any infrared or vibrational spectroscopy technique is the observation of how light is scattered or absorbed upon reaching a material. Commonly, IR wavelength spectroscopy covers wavenumbers ranging from 200 to 4000 cm^-1 Introduction to FTIR Spectroscopy It turns out that these bonds tend to vibrate with frequencies that correspond to infrared-wavelength light, and hence can easily be excited with such a radiation. The choice of IR wavelengths in FTIR method is particularly useful for studying chemical bonds that connect atoms. More specifically, it allows the study of the absorptive and emissive properties of materials. Fourier-transform infrared spectroscopy (or FTIR, for short) is a method of exploring the physical properties of solids, liquids, and gases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
